
1. A system doing work while it heats up has a
A. +w,+q B. +w, -q C. –w, + q D. –w, -q
2. What is the pressure of 1.5 moles of gas in an 8.21L at 100K?
A. 0.5 atm B. 1.0 atm C. 1.5 atm D. 2.0 atm
3. When a 5L gas is compressed from 1 atm to 2.5 atm, what is the new volume?
A. 1.0L B. 1.5L C. 2.0L D. 2.5 L
5. If 0.1 mole of a gas at 300K is put inside a 3.0L vessel, what is P?
A. .0821 B..821 C. 8.21 D. 82.1
6. A sample of air has a vol of 22.5L when the temp is 25C. What is the new V if temp is 100C
A. 29L B. 28.2L C. 19.2 L D. 18.0L
7. At 273K, the sample at 275 mL became 325mL. What is the final temp at C?
A. 323 B. 231 C. 92 D. 50
8. What is P in torr, if vol became 1 L to 0.25L. The initial pressure is 98.0 torr
A. 392 B. 200 C. 24.5 D. 0.75
9. A cylinder has a P of 135 atm at a temp of 20C. If it is heated to 85, what is the new P?
A. 273 B. 165 C. 110 D. 1.00
10. A sample of gas occupies a V oof 0.925L at 25C and 750 mmHg. What is V at STP?
A. 0.981 B. 0.859 C. 0.836 D. 0.742
11. A 50L tank at 0.54 atm and 21C contains refrigerator gas, How many moles are present?
A. 1.12 B. 0.764 C. 0.583 D. 0.0259
II. problem Solving
1. Ge + 1/2O2 GeO H = -255 kJ
Ge + O2 GeO2 H = -534.7 kJ
Determine:.
GeO + 1/2O2 GeO2
2. Give the molecular formula:
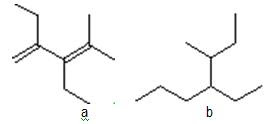
a b
3. determine the functional group of the following
a. fuels c. formic acid
b. acetone d. ethanol
4. give the name of the following;
a. a 5 carbon aldehyde c. a 3 carbon alkyne
b. a 2 carbon alcohol d. a 6 carbon ketone
5. The density of an unknown gas is 2.21 g/L at 1.00 atm and 27.0C. what is the molar mass of this gas?
6. Calculate the mass of NO (30 g/mol) gas occupying 5.2 L at STP.
LONG TEST # 1
I. True or False
1. K of an aqueous solution can be expressed in terms of Kp.
2. Catalysis only affects the rate of reaction.
3. When the concentration of the components is changed, K also changes.
4. Sweating of humans to release body heat is a way of achieving equilibrium.
5. Equilibrium constant is the ratio of the amount of products to the amount of the reactants at equilibrium.
6. The concept of H+ was introduce ed by Arrhenius.
7. The end point is reached when the color of the indicator in titration changes.
8. Baking soda and bleach are acidic; vinegar and orange juice are basic.
II. Problem Solving
1. Given: 2NO2 (brown) N2O4 (colorless). What is the color to be seen in the reaction if: (for a-c)
a. NO2 was added
b. NO2 was removed
c. catalyst was added
d. give the equilibrium constant for the reaction?
e. if [NO2] = 0.05M and [N2O4] = 1 x 10-5 M at equilibrium, what is K?
f. if [NO2] = 0.025M initially, and K is equal to 0.0625, what is the concentration of each species at equilibrium?
2. Complete the table
pH [H+] pOH [OH-] Acidic or basic
| pH | [H+] | pOH | [ | Acidic or basic |
| | | | 6.25 x 10-9 | |
| | | 7.52 | | |
| | 0.0035 | | | |
| 0.5 | | | | |
3. Give the pH at each point. A 32.00 mL of a 0.05 M HCl was titrated with a 0.011M NaOH. a. analyte? b. titrant ? c.0.00 ml d. 62.60 mL e. 145.45 f. 155.34 mL
Labels: Chemical Equilibrium, Exam Help
1. C2H6O(l) + 3O2(g)→2CO2(g) + 3H2O(l)
2C2H4O(l) + 2H2O(l)→2C2H6O(l) + O2(g) H=407KJ
C2H4O(l) + 5/2O2(g)→2CO2(g) + 2H2O(g) H=-1167KJ
2. H2SO4(l)→SO3(g) + H2O(g)
H2SO4(l)→H2S(g) + 2O2(g) H=785KJ
H2S(g) + 2O2(g)→SO3(g) + H2O(l) H=-690KJ
H2O(l)→H2O(g) H=110KJ
QUIZ # 8 III – 1A
1. P1 = 1 atm V1 = 5 L
P2 = 2 atm V2 = ?
2. P1 = 1 atm V1 = 5 L
P2 = ? V2 = 7 L
3. P1 = 6 atm V1 = 5 L
P2 = 2 atm V2 = ?
4. P1 = 6 atm V1 = 5 L
P2 = ? V2 = 2 L
5. P1 = 10 atm V1 = 5 L
P2 = 2 atm V2 = ?
QUIZ # 9 III – 1A
1. T1 = 0C V1 = 5 L
T2 = 10C V2 = ?
2. T1 = 10C V1 = 5 L
T2 = ? V2 = 7 L
3. T1 = 60C V1 = 5 L
T2 = 20C V2 = ?
T2 = ? V2 = 2 L
5. T1 = 25C V1 = 5 L
T2 = -10C V2 = ?
Quiz #1 III – 1A
1. Give the K for: F2 + 2HBr 2HF + Br2
2. Cl2 + 2HBr 2HCl + Br2 , Calc K if [Cl2] = 3M, [HCl] = 5M, [Br2] = 1M, and [HBr] = 2M.
3. What is the conc. of NO if the [NO2] = 1.5M and O2 = 1M and the Kc = 1.1? 2NO + O2 2NO2
QUIZ # 11 III – 1
1. Determine the pressure of a 5.0 mole of gas which occupies 3.5L at 20C.
2. Give the volume occupied by 7.0 moles of gas at STP.
3. Calculate for the moles of a gas which occupies 10L at 100C and 6.5 atm.
4. Determine the temp of a 3.5 mole of gas at 6.0 atm occupying 17L.
Quiz #3 III – 1A
1. Find pH if pOH = 3.4
2. Find pH if [H+] = 3.5 x 10-4
3. Find pOH if pH = 2.1
4. Find pOH if [OH-] = 4.5 x 10-3
5. Find pH if [OH-] = 3.2 x 10-11
6. Find pOH if [H+ ] = 1.0 x 10-12
7. Find [H+] if pOH = 3.2
8. Find [OH-] if pH = 1.8
Labels: Chemistry Lecture, Exam Help
III. Content:
Write the expression for Kc and Kp if applicable, for the following reversible reactions at equilibrium:
1. HF(aq) + H2O (l) <--------> H3O + (aq) + F- (aq)
2. 2 NO(q) + O2 (g) <--------> 2 NO2 (q)
3. 2HgO(s) <--------> 2Hg(l) + O2(g)
4. H2SO3(aq) + H2O(l) <--------> H3O+(aq) + HSO3 (aq)
5. H2(g) + I2(g) <--------> 2HI(g)
III. Content:
1. Consider the following heterogeneous equilibrium:
CaCO3 (s) <--------> CaO (s) + CO2 (g)
At 800 oC, the pressure of CO2 is 0.236 atm. Calculate (a) Kc and (b) Kp for the reaction at
this temperature.
2. The equilibrium constant Kp for the reaction
2 NO2 (g) --------> 2 NO (g) + O2(g)
is 158 at 1000 K. Calculate PO2 if PNO2 = 0.400 atm and PNO = 0.270 atm.
II. Solve the following problems:
1. The equilibrium constant Kp for the reaction
2 NO2 (g) <--------> 2 NO (g) + O2(g)
is 158 at 1000 K. Calculate PO2 if PNO2 = 0.400 atm and and PNO = 0.270 atm.
2. At equilibrium a 1.0 L vessel contains 20.0 mol of H2, 18.0 mol of CO2, 12.0 mol of H2O, and 5.9 mol of CO
at 427°C.What is the value of Kc at this temperature for the following reaction?
CO2(g) + H2(g)<-------->CO(g) + H2O(g)
3. In the reaction: cis-stilbene<-------->trans-stilbene.
The equilibrium constant for this system is 24 at 200 oC. Suppose that initially only cis-stilbene is present at a
concentration of 0.850 mol/L. Calculate the concentrations of these species at equilibrium.
III. Predict the equilibrium shift in the following:
H2 (g) + I2 (g) <--------> 2 HI (g) (change in H > 0)
- addition of H2
- addition of I2
- increase in pressure
- removal of HI
- increase in temperature
III. Content:
I. Identification: Identify the term being described in each of the following statements. Choose the letter of the correct answer on the box below. Use capital letters.
_____ 1. Compounds containing Hydrogen that ionize to yield H+
_____2. Compounds containing Hydrogen that ionize to yield OH-
_____3. An acid that contains 2 ionizable Hydrogen.
_____4. A particle that remains when an acid has donated a Hydrogen ion.
_____5. A substance that can act as both an acid and base.
A. Acid B. Ampoteric C. Base D. Conjugate base E. Conjugate acid F. Monoprotic G. Diprotic
II. Label the Bronsted-Lowry acid and base in the following:
1. H2SO4 + OH <--------> HSO4 - + H2O
2. NH3 + H2O <--------> NH4 + + OH –
3. HSO4 - + H2O <--------> SO4 -2 + H3O +
IV. Answer:
Direction: Solve the following problems.
Show your solution.
1. Calculate the pH
a. H+ = 1.3 x 10 -5
b. H+ = 5.2 x 10 -7
2. Calculate the pOH
a. OH-= 5.3 x 10 -10
b. OH-= 8.9 x 10 -9
3. Calculate the H+
a. OH-= 8.3 x 10 -5
4. Calculate the OH-
a. H+ =9.3 x 10 -5
5. Calculate the H+
a. pH= 3.2
6. Calculate the pH
a. OH-= 3.63 x 10 -6
7. Calculate the pOH
a. pH= 10
8. Calculate the pH
a. 0.76 M KOH
Identify the term/ person/concept being described in each of the following statements/questions.
1. What does pH stands for?
2. Why do we need to compute the Ka value of a weak acid?
3. What does it mean by the higher Ka value?
4. It is a substance that completely dissociate in water, forming H+ and an anion.
5. It is a substance that partially dissociate in water, forming H+ and an anion.
6-9. how do we determine the acid strength of a given weak acid.
10. Who formulated the pH concept?
II. Problem Solving. Answer the following problem carefully. Show your solution.
1. A 0.085 M aqueous solution of a weak, monoprotic acid is 0.85% ionized. Calculate the Ka.
2. The pH of a 0.35 M solution uric acid is 2.17. What is the value of Ka?
III. Answer the following problem carefully. Show your solution.
Give the pH at each point when 100 mL of 0.10 M HCl is titrated with 0.10 M NaOH
a. 0.00 Ml NaOH b. 20 ml NaOH c. 50 mL NaOH d. 100 mL NaOH e. 110 mL
I. True or False: Write T if the statement is True and F if the statement is False.
_____1. When the concentration of the components is changed, K also changes.
_____ 2. The concept of H+ was introduced by Arrhenius.
_____ 3. Equilibrium constant is the ratio of the amount of products to the amount of the reactants at
equilibrium.
_____ 4. The endpoint is reached when the color of the indicator in titration changes.
_____ 5. Catalysis only affects the rate of reaction.
_____ 6. Baling soda and bleach are acidic; vinegar and orange juice are basic.
_____ 7. Sweating of humans to release body heat is a way of achieving equilibrium.
_____ 8. K of an aqueous solution can be expressed in terms of Kp.
_____ 9. A rise in the curve indicates that the titrant is a base while a decline shows that the titrant is an acid.
_____ 10. Strong bases dissociate completely into the cation and OH-.
____ 11. The counting unit in chemistry
____12. It describes the behavior of gases
____13. It is the collision of the molecules of the gases with the conatiner.
____14. it is the result of the molecules collision with one another.
____15. Measurement that deals with the heat changes in the reaction.
____16. quantitative relationships of the reactants and products in a balanced chemical reaction
II. Problem Solving (14 pts.)
![]() 1. Given : 2 NO2 (brown ) N2O4 (colorless). What is the color to be seen in the reaction if :
1. Given : 2 NO2 (brown ) N2O4 (colorless). What is the color to be seen in the reaction if :
(for a-c)
a. NO2 was added
b. NO2 was removed
c. catalyst was added
d. Give the equilibrium constant
e. What is the equilibrium constant (K) if [NO2] = 0.05 M, [N2O4] = 1.0 x 10 -5 M at
equilibrium
f. If NO2 = 0.025 M initially and K = 0.0625. What is the concentration of each species at
equilibrium?
2. Complete the table.(2 pts each)
| pH | H+ | pOH | OH- | Acidic/ Basic |
| (1) | (2) | (3) | 6.25 x 10 -9 | (4) |
| (5) | (6) | 7.52 | (7) | (8) |
| (9) | 0.0035 | (10) | (11) | (12) |
| 0.5 | (13) | (14) | (15) | (16) |
3. Give the pH at each point. A 32 ml of a 0.05 M HCl was titrated with a 0.011 M NaOH.( 5 pts. each pH)
a. analyte
b. titrant
c. 0.00 mL
d. 62.60 mL
e. 145.45 mL
f. 155.34 mL
Labels: Chemistry Lecture
